Article Text
Abstract
Objectives This prospective cohort study aimed to determine the natural history and incidence of oropharyngeal gonorrhoea and chlamydia among a cohort of men who have sex with men (MSM) over a 12-week period, and to examine risk factors associated with incident oropharyngeal infections.
Methods MSM either aged ≥18 years and had a diagnosis of oropharyngeal gonorrhoea by nucleic acid amplification test (NAAT) in the past 3 months or aged 18–35 years who were HIV-negative taking pre-exposure prophylaxis (PrEP) were eligible for this study. Enrolled men were followed up for 12 weeks. Oropharyngeal swabs were collected at week 0 (baseline) and week 12 (end of study). Between these time points, weekly saliva specimens and the number of tongue kissing, penile–oral and insertive rimming partners were collected by post. Oropharyngeal swabs and saliva specimens were tested by NAAT for Neisseria gonorrhoeae and Chlamydia trachomatis. Poisson regression was performed to examine the risk factors (weekly number of partners) associated with incident oropharyngeal gonorrhoea.
Results A total of 100 MSM were recruited. The incidence of oropharyngeal gonorrhoea and chlamydia was 62 (95% CI 37 to 105) and 9 (95% CI 2 to 35)/100 person-years, respectively. The median duration of incident oropharyngeal infection with gonorrhoea was 28 days (IQR=21–36, n=7). The incidence rate ratio (IRR) for oropharyngeal gonorrhoea increased with an increased number of kissing partners (IRR=1.08; 95% CI 1.03 to 1.12) an increased number of penile-oral sex partners (IRR=1.07, 95% CI 1.01 to 1.14) but not with an increased number of insertive rimming partners (IRR=1.11, 95% CI 0.96 to 1.29) or other demographic factors. The IRR and duration of incident oropharyngeal chlamydia were not calculated due to the small number of cases (n=2).
Conclusions MSM have a high incidence of oropharyngeal gonorrhoea and the median duration of infection was less than 3 months.
- chlamydia infections
- epidemiology
- cohort studies
- Neisseria gonorrhoeae
Data availability statement
All data relevant to the study are included in the article or uploaded as supplementary information.
Statistics from Altmetric.com
Introduction
Men who have sex with men (MSM) are disproportionally affected by gonorrhoea, although more recently, cases have also been rising in heterosexuals.1 The rising number of cases is not only expensive for the health system but also increases the probability of Neisseria gonorrhoeae developing resistance.2 This increased potential for resistance is why the World Health Organization (WHO) and the Centers for Disease Control and Prevention (CDC) have described the rises in gonorrhoea and their resistant potential as an urgent health threat.3 4 To combat this threat, both organisations specifically called for improved control of gonorrhoea.
The successful control of gonorrhoea in MSM will be challenging in the current environment because condom use has declined substantially with the introduction of biomedical interventions for HIV control in the late 2010s.1 5 The proportion of Australian MSM who have had condomless anal sex with casual partners in the past 6 months increased significantly from 35% in 2014 to 56% in 2018.1 Accessible healthcare can provide timely diagnosis and treatment for individuals and therefore can reduce ongoing transmission.1 While accessible healthcare works particularly well for symptomatic STIs because the symptoms prompt infected individuals to seek treatment, accessible healthcare is less effective for asymptomatic infections.6 Among heterosexuals, the genitals are the primary site of gonorrhoea infection and these sites are associated with symptoms.7–9 In MSM, the oropharynx or anorectum are the primary sites of infection (sites that are frequently associated with no symptoms),10 suggesting that accessible healthcare will be a less effective control strategy in asymptomatic MSM.
To investigate the difficulty in controlling gonorrhoea in MSM, researchers have proposed new transmission routes.11 12 The findings have highlighted how common gonorrhoea is at the extragenital sites in MSM, particularly at the oropharynx, which has been proposed as an important site for driving onward transmission.11 Further, studies have also shown that both N. gonorrhoeae and Chlamydia trachomatis can be detected in saliva,13 14 suggesting these infections can be transmitted via saliva exchange and activities traditionally considered to be low risk, such as kissing.15 We conducted this longitudinal study to determine the natural history and incidence of oropharyngeal gonorrhoea and chlamydia among a cohort of MSM over a 12-week period to examine risk factors associated with the incident oropharyngeal infections.
Materials and methods
The Gonorrhoea Incidence (GIN) Study was a prospective cohort study of MSM over a 12-week period. Between 15 August 2019 and 30 October 2019, we recruited 100 MSM at the Melbourne Sexual Health Centre (MSHC), Australia. The final visit of the last participant occurred on 29 January 2020. Men were eligible if they had sufficient English language proficiency to understand the study procedure and requirement and were aged ≥18 years and had a diagnosis of oropharyngeal gonorrhoea by nucleic acid amplification test (NAAT) in the past 3 months; or aged 18–35 years who were HIV-negative taking HIV pre-exposure prophylaxis (PrEP); or reported having at least one casual partner in the past 3 months if not taking PrEP. These eligibility criteria were selected because men in these groups were at a higher risk of acquiring gonorrhoea.16 17 Men were not eligible if they were unable to complete the 12-week follow-up, reported ongoing use of antibiotics for 2 weeks or more or were already enrolled in another clinical trial involving the use of antibiotics. MSM living with HIV were also eligible for the study.
The GIN study consisted of two clinic visits over a 12-week period: week 0 (baseline) and week 12 (end of study). To provide increased flexibility, we allowed men to attend the week 12 clinic visit between 11 (minimum) and 14 (maximum) weeks from recruitment. Participants who did not attend the clinic after week 14 were considered as lost to follow-up. Enrolled men were asked to provide clinician-collected oropharyngeal swab and self-collected saliva specimens at weeks 0 and 12, and weekly saliva specimens by the post during the study period. Gonorrhoea and chlamydia screening at all three sites (ie, urethral, oropharyngeal and anorectal) were also offered to all men at week 12. Men who tested positive for N. gonorrhoeae or C. trachomatis at week 0 or 12 were treated at the time of diagnosis according to the standard of care in accordance with the Australian guidelines.18 Men were instructed to self-collect their saliva sample at home weekly from weeks 1 to 11 during the study period and to post the samples to MSHC by mail. A weekly short message service (SMS) was sent to the participant to remind them to send the saliva sample. Participants were advised to accumulate saliva in the mouth for about 30 s and then spit into a specimen jar, then collect the saliva using the UriSwab (Copan Diagnostics, Brescia, Italy). All oropharyngeal swabs and saliva samples were tested by NAAT using Aptima Combo 2 assay (Hologic Panther System; Hologic, San Diego, California, USA). UriSwabs have been successfully used for N. gonorrhoeae or C. trachomatis testing by NAAT in urine and saliva specimens.14 19–21 All oropharyngeal swabs (weeks 0 and 12) were tested in real time, and participants were treated with standard-of-care antibiotics. Saliva samples were stored at −80°C until the end of the study and were batched tested in accordance with the human research and ethics approval for this study; results were not provided to the participants. Participants were required to complete a self-administered paper-based questionnaire each week that asked about the number of men they tongue-kissed, performed penile–oral sex (ie, the participant’s sex partner put his penis in the participant’s mouth) and performed insertive rimming (ie, the participant’s mouth touched and/or licked his partner’s anus) in the past 7 days. The use of any antibiotics and sexual practices in the past 3 months were asked at weeks 0 and 12. All men who tested positive for any STI received standard recommended treatment. Men received an AUD$10 (USD$7) voucher per saliva sample to a maximum of AUD$110 (USD$80) if they sent all saliva samples between weeks 1 and 11 to compensate for their time.
An incident gonorrhoea or chlamydia case was defined as a positive NAAT test from an oropharyngeal swab or saliva sample following a prior negative result during the study period. We excluded week 0 (baseline) positive oropharyngeal gonorrhoea and chlamydia tests, and all these men were treated before they continued in the study. The person-years were calculated from the date of the first sample collected to the date of the last sample. The incidence rate was defined as the number of incident cases per 100 person-years, and the subsequent weeks were excluded if oropharyngeal gonorrhoea continued to be detected. A person could be diagnosed with two incident infections if there were at least two negative results in between positive results. Any equivocal results were considered as positive if there was a positive result in the previous or following week. The duration of an incident infection was defined as the number of days from the date of first positive or equivocal result to the date of the subsequent negative result or the date receiving treatment, excluding men who tested positive only at the week 12 visit. The median duration of incident oropharyngeal chlamydia infections was not calculated due to a small number of incident cases in this cohort (ie, two cases), and we reported each case separately. Poisson regression was performed to examine the risk factors associated with incident oropharyngeal gonorrhoea; the incidence rate ratios (IRRs) and corresponding 95% CIs were reported. Spearman’s correlation was performed between the number of weekly tongue kissing, penile–oral and rimming partners. All statistical analyses were performed using STATA V.14.
Results
A total of 125 men were referred to the research team. We excluded 25 men who were not eligible: 6 did not have any casual partners; 12 were unable to comply with the study protocol requirements due to travel; 4 declined to participate; 2 reported ongoing antibiotics use for 2 weeks or more; and 1 was enrolled in another clinical trial. The remaining 100 men who met the inclusion criteria were enrolled in the study and had a median age of 30 years (IQR=26–33 years). Most participants were HIV-negative men taking PrEP (n=89, 89%), followed by HIV-negative men not taking PrEP (n=9, 9%), with only two men living with HIV (n=2, 2%). There were 22 (22%) men who had a diagnosis of oropharyngeal gonorrhoea 3 months before enrolment. At week 0, men reported a median of 9 (IQR=5–15) male partners in the past 3 months, including a median of 8 (IQR=4–15) tongue-kissing partners, 5 (IQR=3–10) penile–oral sex partners and 2 (IQR=2–6) insertive rimming partners 3 months before enrolment. Ninety-five (95%) men completed the week 12 clinic visit (figure 1). A total of 1162 saliva samples (range of 3–13 per person, including enrolment samples) and 195 oropharyngeal swabs (including enrolment samples) were collected and tested. There was a total of 22 person-years of observation. Men had a median of one (or mean=2) kissing partner, a median of one (or mean=2) oral sex partner and a median of zero (or mean=1) insertive rimming partner each week during the study. Each of these three practices were strongly correlated with each other with correlation coefficients of between 0.50 and 0.80 (see online supplemental figure 1). The concordance for the saliva sample and swab specimens was relatively high at enrolment but less so at week 12 (see online supplemental tables 1–4).
Supplemental material
Supplemental material
Supplemental material
Supplemental material
Supplemental material
Study flowchart. GIN, Gonorrhoea Incidence; MSM, men who have sex with men. Note. 75 men returned 11 weekly swabs OR returned 10 swabs because they attended for their final visit in week 11 due to scheduling their 3 month visit as permitted.
At week 0, 16 men (16%, 95% CI 9% to 25%) tested positive for N. gonorrhoeae and 3 (3%, 95% CI 1% to 9%) tested positive for C. trachomatis from either the oropharyngeal swabs or saliva samples. Two (95% CI 0% to 7%) men had infectious syphilis diagnosed at week 0. All infections detected at week 0 were treated and men only contributed person-time to the overall incidence rates post-treatment.
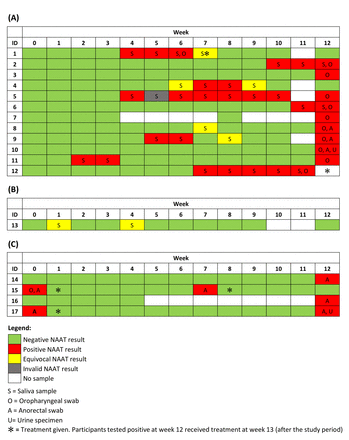
During the 12 weeks of follow-up, there were 14 incident oropharyngeal gonorrhoea diagnoses in 12 men, and the overall incidence of oropharyngeal gonorrhoea was 62.43 (95% CI 36.97 to 105.41) per 100 person-years. Of the 14 incident infections, 8 were detected on weekly saliva samples (between weeks 1 and 11), and 4 cases were only detected on the oropharyngeal swabs at week 12 (figure 2). Eight men had infection detected on saliva samples between weeks 1 and 11. One man collected his saliva sample only 3 days before his week 12 visit, and therefore, he was excluded from the calculation of the duration of incident infection (participant 6, figure 2). The median duration that incident oropharyngeal gonorrhoea persisted for was 28 days (IQR=21–36, n=7) (figure 3).
Incident gonorrhoea cases among (A) 12 men tested positive for Neisseria gonorrhoeae in the saliva or the oropharynx; (B) 1 man with equivocal results for N. gonorrhoeae in the saliva; and (C) 4 men tested positive for N. gonorrhoeae at other anatomical sites except for the oropharynx. ID, identification number; NAAT, nucleic acid amplification test. Note. The ID numbers were recorded forpublication and they were not the actual study ID. Participant 1 had anoropharyngeal swab taken at week 6 as he presented to the clinic as a contactof infection. He presented with symptoms including sore throat, dysuria andclear urethral discharge. He was tested positive for oropharyngeal gonorrhoeaand urethral Mycoplasmagenitalium. Participant 15 had an anorectalswab at week 7 and tested positive for anorectal gonorrhoea. He presented tothe clinic as a contact of infection but did not have any symptoms on the day.
Kaplan-Meier curve of the duration of incident oropharyngeal gonorrhoea infection. CI, confidence intervals.
The IRR for oropharyngeal gonorrhoea increased with increased number of kissing partners (IRR=1.08, 95% CI 1.03 to 1.12, p=0.001) and increased number of penile–oral sex partners (IRR=1.07; 95% CI 1.01 to 1.14; p=0.016) but not with increased number of insertive rimming partners (IRR=1.11, 95% CI 0.95 to 1.29, p=0.175) or other demographic factors (table 1). Of the 14 incident oropharyngeal gonorrhoea cases, all men (100%) reported kissing in prior week, while two (14%) had kissing only with no penile–oral sex and insertive rimming; six (43%) had kissing and penile–oral sex but no rimming; and the remaining six (43%) had kissing, penile–oral sex and rimming. There were 69 men reporting 244 weekly intervals with no kissing partners, of which 15 weekly intervals were with penile–oral sex partners and 4 weekly intervals were with insertive rimming partners; however, none tested positive for N. gonorrhoeae in the saliva or oropharynx. In contrast, there were 80 men reporting 349 weekly intervals with no penile–oral sex partners, of which 120 weekly intervals were with kissing partners and 27 weekly intervals were with insertive rimming partners; 2 men tested positive for N. gonorrhoeae in the saliva or oropharynx. Furthermore, there were 92 men reporting 683 weekly intervals with no insertive rimming partners, of which 94 weekly intervals were with kissing partners and 12 weekly intervals were with penile–oral sex partners; 6 men tested positive for N. gonorrhoeae in the saliva or oropharynx, 1 of whom had a second incident infection.
Factors associated with incidence of throat gonorrhoea (incidence defined as one event in the case of a run of events)
There were two incident oropharyngeal chlamydia diagnoses in two men, and the overall incidence of oropharyngeal chlamydia was 8.85 (95% CI 2.21 to 35.39) per 100 person-years. Of the two incident infections, both were detected on weekly saliva samples: one was detected at week 4 and then tested negative at week 5, and the second was tested positive on three consecutive saliva samples from weeks 10–12 as well as his week 12 oropharyngeal swab.
There were 7 cases of incident anorectal gonorrhoea, 2 cases of incident urethral gonorrhoea (figure 3), 10 cases of incident anorectal chlamydia, 5 cases of incident urethral chlamydia and 3 cases of infectious syphilis (2 primary and 1 early latent).
Discussion
We estimated that incidence of oropharyngeal infection in this cohort was more than 60% per year, and about half of the cases were detected first on saliva samples between week 0 and week 12. Furthermore, the estimated median duration of incident oropharyngeal gonorrhoeal infection was 28 days, consistent with past studies showing oropharyngeal gonorrhoeal infection has a relatively short duration.22 23 Incident oropharyngeal gonorrhoea was associated with exposure to a partner’s mouth (ie, kissing) or penis (ie, penile–oral sex) but not their partner’s anus (ie, rimming) in the previous week. Unfortunately, separating the independent contribution of the different exposures is challenging because exposure to these three sites commonly occurs together in the same sexual act.24 25 There has been only one similar study published in abstract form that reported a high incidence of oropharyngeal gonorrhoea of about 35% per year based on weekly oropharyngeal swab sampling; however, no data on sexual risk were provided.22 Our data suggest that the true incidence of oropharyngeal gonorrhoea is higher than what has been previously appreciated even when screening occurs every 3 months. When considering the number of exposures to different mouths, penises and anuses and the prevalence of gonorrhoea at each site, it is challenging to see how penises alone could be responsible for this high incidence of oropharyngeal gonorrhoea.11
Our study had several limitations. First, we used saliva samples that were sent through the postal service to avoid the men needing to return to the clinic every week for an oropharyngeal swab. This strategy did allow us to have a very high return rate (95%), but saliva appeared to be considerably less sensitive for oropharyngeal gonorrhoea than a throat swab using the Aptima combo that tests for RNA. However, our previous studies using DNA detection suggested saliva testing for gonorrhoea was quite sensitive for the detection of N. gonorrhoeae and comparable with the detection using oropharyngeal swabs.13 This suggests that it is likely that we have underestimated the true incidence and duration of infection for oropharyngeal gonorrhoea in this study. Further studies are required to determine the optimal testing and sampling for saliva samples for the detection of N. gonorrhoeae. Second, the study was conducted around winter and spring seasons. STI cases peak around summer, and therefore, we might have underestimated the incidence due to the seasonal variations of STI and sexual practices.26 27 Third, we specifically chose men who were likely to have a high incidence of oropharyngeal gonorrhoea, and thus, our estimate may not be generalised to the entire MSM population. Our population, for example, had nine partners in the past 3 months, which is about three times higher than our entire clinic population, which is about three partners every 3 months.28 We estimated that the incidence of urethral gonorrhoea (8/100 person-years) and syphilis (12/100 person-year) in our study was similar to that of a large study of MSM taking PrEP in Melbourne (the “PrEPX” study).29 However, the estimated incidence of oropharyngeal gonorrhoea in the present study (55/100 person-year) was nearly threefold higher than the PrEPX study (20/100 person-years) and suggests that cases in the PrEPX study were missed because sampling occurred only every 3 months. Finally, 4 out of 12 men had their incident oropharyngeal N. gonorrhoeae infection detected at the week 12 visit only, and so we might have underestimated the duration of the infection’s persistence due to the short follow-up time of 12 weeks in our study.
To our best knowledge, there has been only one study sampling MSM weekly for the detection of N. gonorrhoeae in the oropharynx that was conducted in the USA.22 The study recruited 140 men who self-collect oropharyngeal swabs at home over a 48-week period, but only 48 (34%) men were included in the final analysis. The study estimated the incidence of oropharyngeal gonorrhoea was 35/100 person-years, which is lower than the estimate in our study. Both Barbee’s and our studies report an incidence of oropharyngeal gonorrhoea that is substantially higher than the incidence seen in other studies, including cohort studies of PrEP participants. Furthermore, Barbee et al22 estimated that the median duration of oropharyngeal gonorrhoea infection was 9 weeks (95% CI 3 to 19), which was longer than our estimate of 4 weeks. The difference between the two studies is likely due to the limited sensitivity of posted saliva samples tested by RNA in our study. However, both Barbee’s and our study do suggest that the incidence of oropharyngeal gonorrhoea is substantially higher than what has previously been appreciated and that some infections are short-lived and would have been missed with 3 monthly screenings.22
Our study raises questions about the currently accepted routes of transmission for gonorrhoea that dictate that oropharyngeal gonorrhoea is predominantly acquired from urethral infection following oral sex. In our study, oropharyngeal gonorrhoea was associated with exposure in the previous week to both a partner’s mouth or penis but not the anus. This is consistent with a previous study showing having kissing-only partners is an independent risk factor for oropharyngeal gonorrhoea.15 We note that separating the individual contribution is statistically impossible when collinearity is so high. However, we did find that there were no oropharyngeal cases in men with no kissing partners in the previous week, yet among men with no oral sex partners, six oropharyngeal infections occurred and among men with no rimming partners, seven oropharyngeal cases occurred. We also had only two cases of urethral gonorrhoea occur in the men, and both presented with urethral symptoms, suggesting that the prevalence of urethral gonorrhoea in men like the ones in our study would be very low. In contrast, the proportion of men who had oropharyngeal gonorrhoea detected in the oropharynx at week 0 or 12 was more than 10%, indicating that the urethra is unlikely to be the sole source of the high incidence of oropharyngeal gonorrhoea we observed.
We found a substantially lower incidence of chlamydial infection in the oropharynx of only about 9/100 person-years despite a considerable incidence of urethral (20/100 person-years) and particularly anorectal infection (40/100 person-years), although the latter two sites were solely based on samples taken at week 12. In Barbee’s study, they collected weekly oropharyngeal and anorectal samples for chlamydia and found an incidence of anorectal chlamydia of 59/100 years but the incidence of oropharyngeal chlamydia was not reported.22 Taken together, these data suggest that the oropharynx plays a substantially less important role for chlamydia than for gonorrhoea.
To conclude, our results suggested that the incidence of oropharyngeal gonorrhoea was relatively high among MSM predominately taking PrEP and that the duration of infection was short and, some persisted for only a few weeks. This suggests that a 3-monthly screening approach may not be sufficient to control gonorrhoea. The short duration of infection implies that if screening were to be effective, it would need to be very frequent. However, we showed that a considerable number of the infections cleared spontaneously and did not require treatment. Before any recommendation could be made on more frequent screening, further studies would be required to delineate the harms, benefits, cost-effectiveness and adherence to such frequent screening intervals.
Key messages
Men who have sex with men have a high incidence of oropharyngeal gonorrhoea of 62/100 person-years.
The median duration of oropharyngeal gonorrhoea infection was short (ie, 28 days).
The incidence oropharyngeal gonorrhoea was associated with an increased number of kissing partners, penile–oral sex partners but not with insertive rimming partners.
Data availability statement
All data relevant to the study are included in the article or uploaded as supplementary information.
Ethics statements
Patient consent for publication
Ethics approval
Written consent was obtained from all participants before commencing the study. This study was approved by the Alfred Hospital Ethics Committee (272/19).
References
Supplementary materials
Supplementary Data
This web only file has been produced by the BMJ Publishing Group from an electronic file supplied by the author(s) and has not been edited for content.
Footnotes
EPFC and LAV are joint first authors.
Handling editor Jonathan Ross
Twitter @EricPFChow, @lenkavod, @drdebwilliamson, @JHocking01, @BioMinnie, @Cat_Bradshaw_, @kitfairley
Correction notice This article has been corrected since it was first published online. Table 1 has been updated.
Contributors EPFC, LAV and CKF conceived and designed the study. EPFC, LAV, KM and CKF designed the study materials. LAV and KM oversaw the ethics approval and study procedures. KM was involved in database management, specimen collection, processing and storage, and performed chart review. LAV performed the statistical analyses. EPFC and JSH assisted with the statistical analyses. EPFC, LAV and CKF wrote the first draft of the manuscript. DW oversaw the laboratory testing results and was assisted by VDP. All authors were involved in revising the manuscript for important intellectual content and approved the final version.
Funding This trial was supported by an Australian National Health and Medical Research Council (NHMRC) Project Grant (No. 568971). CKF and CSB are supported by an NHMRC Leadership Investigator Grant (GNT1172900 and GNT1173361, respectively). DW and EPFC are supported by an Australian NHMRC Emerging Leadership Investigator Grant (GNT1174555 and GNT1172873, respectively). JSH is supported by an NHMRC Senior Research Fellowship (GNT1136117).
Competing interests None declared.
Provenance and peer review Not commissioned; externally peer reviewed.
Supplemental material This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.